
|
Developed by 

|
Supported by 


|
LAPSCOVERY (Long Acting Protein / Peptide Discovery Platform Technology)
Developer(s)

|
Hanmi Pharmaceutical Originator
https://hanmipharm.com/
South Korea Hanmi Pharmaceutical Ltd., founded in 1973 in South Korea, is a leading R&D-driven company specializing in innovative drug development. It pioneers platform technologies like LAPSCOVERY for long-acting biologics and ORASCOVERY for oral chemotherapeutics. Hanmi collaborates with global firms, including Sanofi, Eli Lilly, and Genentech, advancing oncology, diabetes, and rare disease treatments. |
Sponsor(s)
|
No sponsor indicated |
Partnerships

|
Merck Sharp & Dohme https://www.msd.com/ |
Technology information
Type of technology
aglycosylated Fc based particles
Administration route
Intravenous, Subcutaneous, Intramuscular
Development state and regulatory approval
efocipegtrutide (GLP-1 triple agonist )
Phase II
Efocipegtrutide Fast Track designation and orphan drug status for the treatment of nonalcoholic steatohepatitis (MASH).
Description
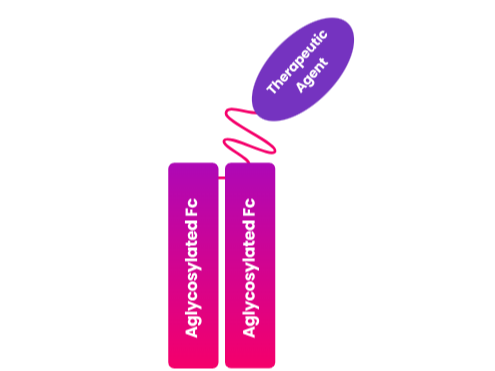
LAPSCOVERY is a long-acting platform technology designed to extend the half-life of biologics administered systemically. The API (therapeutic proteins) is fused with aglycosylated Fc fragments, which enhances the pharmacokinetics of the API. The monomeric Fc fragments help to reduce receptor-mediated clearance, thereby delaying renal filtration and prolonging the therapeutic effect.
Technology highlight
1. The flexible linker minimizes loss of intrinsic activity 2. Highest bioavailability reduces dose level 3. Low Immunogenicity 4. Minimal Loss of activity 5. Increased cellular solubility 6. No loss of neonatal Fc receptor (FcRn) binding
Technology main components
1. Two Aglycolated Fc Fragments 2. Carrier (e.g., Ethylene vinyl acetate) 3. Non-peptide biocompatible polymer (Linker) 4. Excipients—lactose , dextrose , sucrose , sorbitol, mannitol , xylitol , erythritol , maltitol , starch , acacia rubber, alginate , gelatin , calcium phosphate , calcium silicate , cellulose , methyl cellulose , microcrystalline cellulose , polyvinylpyrrolidone, water , methyl hydroxybenzoate , propyl hydroxybenzoate , talc , magnesium stearate , mineral oil , etc. 5. Fillers, anti-coagulant , lubricants, humectants, flavoring agents, preservatives, etc.
Information on the raw materials sourcing, availability and anticipated price
Not provided
Delivery device(s)
Single-dose prefilled syringe
APIs compatibility profile
API desired features
Small molecules
LAPSCOVERY specially focuses on protein-based small molecules designed to target epidermal growth factor receptors (EGFR), including HER2 and HER4, with compounds such as poziotinib,Tuspetinib.
Proteins
LAPSCOVERY targets a diverse range of proteins, including insulin homologues, human growth hormone, GLP-1 agonists, glucagon, granulocyte colony-stimulating factor, interleukins, and bispecific antibodies.
Additional solubility data
Not provided
Additional stability data
Not provided
API loading: Maximum drug quantity to be loaded
50-75 wt%
API co-administration
2 different APIs : Not provided
LogP
Not provided
Not provided
Scale-up and manufacturing prospects
Scale-up prospects
Hanmi has a bio plant in Pyeongtaek for the LAPSCOVERY-based biologics occupying a 4,600 m² area.
Tentative equipment list for manufacturing
Not provided
Manufacturing
Not provided
Specific analytical instrument required for characterization of formulation
Not provided
Clinical trials
HM-FLUT-301
Identifier
NCT02941679
Link
https://clinicaltrials.gov/study/NCT02941679
Phase
Phase III
Status
Unknown status
Sponsor
Hanmi Pharmaceutical Company Limited
More details
A phase 3 study to evaluate efficacy and safety of HCP1202
Purpose
Clinical Efficacy and Safety Evaluation of HCP1202 in COPD Patients
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2016-10-01
Anticipated Date of Last Follow-up
2016-10-19
Estimated Primary Completion Date
2018-02-01
Estimated Completion Date
2018-04-01
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: * Male or Female adults aged ≥ 40 years. * Patients diagnosed with COPD. * Patients with FEV1/FVC \< 0.7 at screening. * Patients with a post-bronchodilator FEV1 \< 60% of the predicted normal OR Patients with a post-bronchodilator FEV1 \< 80% of the predicted normal if COPD exacerbation is moderate or worse developed at least twice within the past year or hospitalization occurred at least once within the past year due to COPD exacerbation. * Patients with COPD Assessment Test ≥ 10. * Patients with a history (current or ex-smokers) of smoking 10 pack-years or more (e.g. 10 pack years = 1 pack/day x 10 years, or ½ pack/day x 20 years). * Patients who understand the process of clinical trial and signed written informed consent. Exclusion Criteria: * Patients with a cur
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
252
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-M
Identifier
NCT03353350
Link
https://clinicaltrials.gov/study/NCT03353350
Phase
Phase III
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
Primary Objective: To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo in glycated hemoglobin (HbA1c) change in participants with T2DM (Type 2 Diabetes Mellitus) inadequately controlled with diet and exercise. Secondary Objectives: * To demonstrate the superiority of once-weekly injection of efpeglenatide in comparison to placebo on glycemic control * To demonstrate the superiority of once-weekly injection of efpeglenatide in comparison to placebo on body weight * To evaluate the safety of once-weekly injection of efpeglenatide
Purpose
Efficacy and Safety of Efpeglenatide Versus Placebo in Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Diet and Exercise
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2017-12-05
Anticipated Date of Last Follow-up
2022-01-11
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-01-29
Actual Completion Date
2020-09-07
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * Participants must be at least 18 years of age at the time of signing the informed consent. * Participants with T2DM, and treated with diet and exercise. * Hemoglobin A1c between 7.0% and 10.0% (inclusive) measured by the central laboratory at Screening. Exclusion criteria: * Clinically relevant history of gastrointestinal disease associated with prolonged nausea and vomiting, including (but not limited to) gastroparesis, unstable and not controlled gastroesophageal reflux disease within 6 months prior to Screening or history of surgery affecting gastric emptying. * History of pancreatitis (unless pancreatitis was related to gallstone and cholecystectomy has been performed) and pancreatitis during previous treatment with incretin therapies, chronic pancreatitis, and
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
406
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-LIL2-101
Identifier
NCT06724016
Link
https://clinicaltrials.gov/study/NCT06724016
Phase
Phase I
Status
Recruiting
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This is a First-in-Human, Phase 1, Dose-Escalation and Dose-Expansion study of HM16390, as a single agent to assess safety, tolerability, MTD, RP2D, PK, and efficacy in patients with advanced or metastatic solid tumors. Dose-Escalation Part is planned to establish the MTD or RDs for the randomized Dose-Ranging Part. Based on the results of the Dose-Escalation Part, additional eligible subjects will be randomized 1:1 into at each dose level. After a comprehensive review of available data from both Dose-Escalation Part and Dose-Ranging Part, the potential RP2D to be tested in the Dose-Expansion Part is determined. Dose-Expansion Part is designed to assess the potential efficacy of HM16390 as a single agent when administered at the potential RP2D to subjects in indication-specific expansion
Purpose
Dose Escalation and Expansion Study of HM16390 in Advanced or Metastatic Solid Tumors
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
2024-12-11
Actual Start Date
Not provided
Anticipated Date of Last Follow-up
2024-12-11
Estimated Primary Completion Date
2030-03-01
Estimated Completion Date
2030-03-01
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Key Inclusion Criteria: * Have a histologically and/or cytologically confirmed advanced or metastatic solid tumor and have failed or are intolerant to standard therapy with clinical benefit. * Patients in the Dose-Escalation Part must have evaluable or measurable disease at baseline and the patients for Dose-Ranging and Dose-Expansion Part must have at least one measurable lesion at baseline by computed tomography (CT) or magnetic resonance imaging (MRI) per Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. * Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1. * Age of 18 years or older (or country's legal age of majority if the legal age was \>18 years) * Adequate renal function. * Adequate hematologic function. * Adequate liver function. Key Exclusion Crit
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
215
Allocation
Not provided
Intervention model
Single group assignment
Intervention model description
Not provided
Masking
Open label
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-FLUT-301
Identifier
NCT02941679
Link
https://clinicaltrials.gov/study/NCT02941679
Phase
Phase III
Status
Unknown status
Sponsor
Hanmi Pharmaceutical Company Limited
More details
A phase 3 study to evaluate efficacy and safety of HCP1202
Purpose
Clinical Efficacy and Safety Evaluation of HCP1202 in COPD Patients
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2016-10-01
Anticipated Date of Last Follow-up
2016-10-19
Estimated Primary Completion Date
2018-02-01
Estimated Completion Date
2018-04-01
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: * Male or Female adults aged ≥ 40 years. * Patients diagnosed with COPD. * Patients with FEV1/FVC \< 0.7 at screening. * Patients with a post-bronchodilator FEV1 \< 60% of the predicted normal OR Patients with a post-bronchodilator FEV1 \< 80% of the predicted normal if COPD exacerbation is moderate or worse developed at least twice within the past year or hospitalization occurred at least once within the past year due to COPD exacerbation. * Patients with COPD Assessment Test ≥ 10. * Patients with a history (current or ex-smokers) of smoking 10 pack-years or more (e.g. 10 pack years = 1 pack/day x 10 years, or ½ pack/day x 20 years). * Patients who understand the process of clinical trial and signed written informed consent. Exclusion Criteria: * Patients with a cur
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
252
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-101
Identifier
NCT03374241
Link
https://clinicaltrials.gov/study/NCT03374241
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
Single ascending dose of HM15211 in healthy obese subjects.
Purpose
A First-in-human Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of HM15211(Efocipegtrutide)
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-04-04
Anticipated Date of Last Follow-up
2025-01-23
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2018-09-14
Actual Completion Date
2018-09-14
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Female subjects must be non-pregnant and non-lactating Exclusion Criteria: * Participation in an investigational study within 30 days prior to dosing
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
41
Allocation
Randomized
Intervention model
Sequential assignment
Intervention model description
Not provided
Masking
Triple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-102
Identifier
NCT03744182
Link
https://clinicaltrials.gov/study/NCT03744182
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This study is a phase 1 study to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of multiple doses of HM15211 in obese subjects with NAFLD
Purpose
A Study of Multiple Doses of HM15211(Efocipegtrutide) in Obese Subjects With NAFLD
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-11-01
Anticipated Date of Last Follow-up
2025-01-23
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-03-18
Actual Completion Date
2020-03-18
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Body mass index ≥ 30 kg/m2 * Waist circumference ≤ 57 inches * Fasting Plasma Glucose \< 7 mmol/L (126 mg/dL) * HbA1c \< 6.5% * Controlled Attenuation Parameter ≥ 300 dB/m by FibroScan * Liver fat by MRI-PDFF ≥ 10%. Exclusion Criteria: * A history of or active chronic liver disease due to alcohol, auto-immune, HIV, HBV or active HCV-infection or NASH disease * Any history of clinically significant chronic liver disease including esophageal varices, ascites, encephalopathy or any hospitalization for treatment of chronic liver disease; or Model for End Stage Liver Disease (MELD) ≥ 10 * Previous surgical treatment for obesity * Uncontrolled hypertension * Any weight control treatment * History or current diagnosis of acute or chronic pancreatitis or factors for pancre
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
66
Allocation
Randomized
Intervention model
Sequential assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-201
Identifier
NCT04505436
Link
https://clinicaltrials.gov/study/NCT04505436
Phase
Phase II
Status
Recruiting
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This study is a phase 2 study to Evaluate Efficacy, Safety and Tolerability of HM15211 Treatment for 12 Months in Subjects with Biopsy Confirmed NASH
Purpose
Study to Evaluate Efficacy, Safety and Tolerability of HM15211(efocipegtrutide) in Subjects
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2020-07-31
Anticipated Date of Last Follow-up
2025-01-20
Estimated Primary Completion Date
2025-05-11
Estimated Completion Date
2025-11-10
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: * United States Sites: Adults ≥ 18 to ≤ 70 years. * Korean Sites: Adults ≥ 19 to ≤ 70 years. * BMI ≥ 18 kg/m2, with stable body weight (defined as change \< 5%) by history for 3 months prior to screening or since baseline liver biopsy, whichever is earlier. * Subjects have a diagnosis of noncirrhotic NASH with liver fibrosis (Fibrosis stage F1-F3) confirmed by liver biopsy within 6 months of Day -7. * MRI-PDFF performed at screening with ≥ 8% steatosis. Exclusion Criteria: * Subjects with a history of active or chronic liver disease, including alcoholic liver disease, viral hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, autoimmune hepatitis, Wilson's disease, hemochromatosis, alpha-1 antitrypsin deficiency, human immunodeficiency virus (HIV). *
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
240
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
6024-014
Identifier
NCT06052566
Link
https://clinicaltrials.gov/study/NCT06052566
Phase
Phase I
Status
Completed
Sponsor
Merck Sharp & Dohme LLC
More details
The purpose of this study is to evaluate the pharmacokinetics of efinopegdutide in participants with hepatic impairment compared to healthy participants, and to examine the safety and tolerability of efinopegdutide.
Purpose
A Study of Efinopegdutide in Participants With Hepatic Impairment (MK-6024-014)
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2023-11-21
Anticipated Date of Last Follow-up
2024-12-13
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2024-12-05
Actual Completion Date
2024-12-05
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
The main inclusion and exclusion criteria include but are not limited to the following: Inclusion Criteria: * A participant assigned female at birth is eligible to participate if not pregnant or breastfeeding, is not a participant of childbearing potential (POCBP), or is a POCBP and agrees to follow contraceptive guidance during the study intervention period and for at least 35 days after the last dose of study intervention. * For participants with moderate or severe hepatic impairment: Have a diagnosis of chronic (\>6 months), stable, hepatic impairment with features of cirrhosis due to any etiology (stability of hepatic disease should correspond to no acute episodes of illness within the previous 2 months due to deterioration in hepatic function). Exclusion Criteria: * History of can
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
22
Allocation
Not provided
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Open label
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
6024-016
Identifier
NCT06482112
Link
https://clinicaltrials.gov/study/NCT06482112
Phase
Phase II
Status
Not provided
Sponsor
Merck Sharp & Dohme LLC
More details
This study will evaluate the effect of efinopegdutide administration once every 2 weeks (Q2W) versus once weekly (Q1W) on mean relative reduction from baseline in liver fat content (LFC) after 28 weeks, as well as the safety and tolerability of the different regimens of efinopegdutide.
Purpose
Alternate Dosing Study of MK-6024 in Adults With Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) (MK-6024-016)
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2024-07-29
Anticipated Date of Last Follow-up
2024-11-20
Estimated Primary Completion Date
2025-06-23
Estimated Completion Date
2025-06-23
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: The main inclusion criteria include but are not limited to the following: * Has body mass index (BMI) ≥25 kg/m\^2 (≥23 kg/m\^2 for Asian participants) AND has stable weight, defined as ≤5% gain or loss of body weight for at least 3 months before screening * Has no history of type 2 diabetes mellitus (T2DM) OR has a history of T2DM with a glycated hemoglobin (A1C) ≤9% AND the T2DM is controlled by diet or stable doses of oral antihyperglycemic agents (AHAs) Exclusion Criteria: The main exclusion criteria include but are not limited to the following * Has a history or evidence of chronic liver disease other than Metabolic dysfunction-associated steatotic liver disease (MASLD) or Metabolic dysfunction-associated steatohepatitis (MASH) * Has evidence of decompensated l
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
129
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Open label
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-O
Identifier
NCT03496298
Link
https://clinicaltrials.gov/study/NCT03496298
Phase
Phase III
Status
Terminated
Sponsor
Sanofi
More details
Primary Objective: To demonstrate that efpeglenatide 4 and 6 mg was noninferior to placebo on 3-point major adverse cardiac events (MACE) in Type 2 diabetes mellitus (T2DM) participants at high cardiovascular (CV) risk. Secondary Objectives: To demonstrate that efpeglenatide 4 and 6 mg was superior to placebo in T2DM participants with high CV risk on the following parameters: * 3-point MACE. * Expanded CV outcome. * Composite outcome of new or worsening nephropathy. To assess the safety and tolerability of efpeglenatide 4 and 6 mg, both added to standard of care in T2DM participants at high CV risk.
Purpose
Effect of Efpeglenatide on Cardiovascular Outcomes
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-04-27
Anticipated Date of Last Follow-up
2021-09-16
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-12-10
Actual Completion Date
2020-12-10
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * T2DM with glycosylated hemoglobin (HbA1c) greater than (\>) 7 percentage. * Age 18 years or older who met at least one of the cardiovascular disease criteria or age 50 years (male), 55 years (female) or older with glomerular filtration rate greater than or equal to 25 and less than 60 milliliters per minute and at least had one cardiovascular risk factor. * Female participants agreed to follow contraceptive guidance. * Signed written informed consent. Exclusion criteria: * Clinically relevant history of gastrointestinal disease associated with prolonged nausea and vomiting. * History of chronic pancreatitis or acute idiopathic pancreatitis or diagnosis of any type of acute pancreatitis within 3 months prior to screening. * Personal or family history of medullary th
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
4076
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-M
Identifier
NCT03353350
Link
https://clinicaltrials.gov/study/NCT03353350
Phase
Phase III
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
Primary Objective: To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo in glycated hemoglobin (HbA1c) change in participants with T2DM (Type 2 Diabetes Mellitus) inadequately controlled with diet and exercise. Secondary Objectives: * To demonstrate the superiority of once-weekly injection of efpeglenatide in comparison to placebo on glycemic control * To demonstrate the superiority of once-weekly injection of efpeglenatide in comparison to placebo on body weight * To evaluate the safety of once-weekly injection of efpeglenatide
Purpose
Efficacy and Safety of Efpeglenatide Versus Placebo in Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Diet and Exercise
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2017-12-05
Anticipated Date of Last Follow-up
2022-01-11
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-01-29
Actual Completion Date
2020-09-07
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * Participants must be at least 18 years of age at the time of signing the informed consent. * Participants with T2DM, and treated with diet and exercise. * Hemoglobin A1c between 7.0% and 10.0% (inclusive) measured by the central laboratory at Screening. Exclusion criteria: * Clinically relevant history of gastrointestinal disease associated with prolonged nausea and vomiting, including (but not limited to) gastroparesis, unstable and not controlled gastroesophageal reflux disease within 6 months prior to Screening or history of surgery affecting gastric emptying. * History of pancreatitis (unless pancreatitis was related to gallstone and cholecystectomy has been performed) and pancreatitis during previous treatment with incretin therapies, chronic pancreatitis, and
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
406
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-D
Identifier
NCT03684642
Link
https://clinicaltrials.gov/study/NCT03684642
Phase
Phase III
Status
Terminated
Sponsor
Sanofi
More details
Primary Objective: To demonstrate the non-inferiority of once weekly injection of efpeglenatide in comparison to once weekly injection of dulaglutide on glycated hemoglobin (HbA1c) change in participants with Type 2 diabetes mellitus (T2DM) inadequately controlled with metformin. Secondary Objectives: * To demonstrate the superiority of once weekly injection of efpeglenatide with once weekly injection of dulaglutide on glycemic control. * To demonstrate the superiority of once weekly injection of efpeglenatide with once weekly injection of dulaglutide on body weight. * To evaluate the safety of once weekly injection of efpeglenatide and once weekly injection of dulaglutide.
Purpose
Efficacy and Safety of Efpeglenatide Versus Dulaglutide in Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Metformin
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-09-26
Anticipated Date of Last Follow-up
2021-10-28
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-10-13
Actual Completion Date
2020-11-17
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * Participant must be greater than or equal to (\>=) 18 years of age at the time of signing the informed consent. * Participants with T2DM. * Diabetes diagnosed at least 1 year before screening. * Participants on stable dose of at least 1500 milligram per day (mg/day) of metformin, or tolerated maximum dose, or as per country regulation if less, for at least 3 months prior to screening. * HbA1c between 7.0 percent (%) and 10.0% (inclusive) measured by the central laboratory at screening. Exclusion criteria: * Retinopathy or maculopathy with one of the following treatments, either recent (within 3 months prior to screening) or planned: intravitreal injections or laser or vitrectomy surgery. * Clinically relevant history of gastrointestinal (GI) disease associated with
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
908
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Open label
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-S
Identifier
NCT03770728
Link
https://clinicaltrials.gov/study/NCT03770728
Phase
Phase III
Status
Terminated
Sponsor
Sanofi
More details
Primary Objective: To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo in glycated hemoglobin (HbA1c) change in participants with type 2 diabetes mellitus (T2DM) inadequately controlled with metformin alone or in combination with sulfonylurea (SU). Secondary Objectives: * To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo on glycemic control. * To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo on body weight. * To evaluate the safety of once weekly injection of efpeglenatide.
Purpose
Efficacy and Safety of Efpeglenatide Versus Placebo in Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Metformin Alone or in Combination With Sulfonylurea
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2019-08-01
Anticipated Date of Last Follow-up
2021-11-04
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-11-28
Actual Completion Date
2020-12-27
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * Participant must be greater than or equal to (\>=)18 years of age at the time of signing the informed consent. * Participants with T2DM. * Diabetes diagnosed at least 1 year before screening. * Participants on metformin alone or in combination with SU, for at least 3 months prior to screening. * Glycated hemoglobin between 7.0% and 10.0% (inclusive) measured by the central laboratory at screening. Exclusion criteria: * History of severe hypoglycemia requiring emergency room admission or hospitalization within 3 months prior to screening. * Retinopathy or maculopathy with one of the following treatments, either recent (within 3 months prior to screening) or planned: intravitreal injections or laser or vitrectomy surgery. * Clinically relevant history of gastrointest
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
312
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
AMPLITUDE-L
Identifier
NCT03713684
Link
https://clinicaltrials.gov/study/NCT03713684
Phase
Phase III
Status
Terminated
Sponsor
Sanofi
More details
Primary Objective: To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo in glycated hemoglobin (HbA1c) change in participants with type 2 diabetes mellitus (T2DM) inadequately controlled with basal insulin alone or in combination with oral antidiabetic drugs (OADs). Secondary Objectives: * To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo on glycemic control. * To demonstrate the superiority of once weekly injection of efpeglenatide in comparison to placebo on body weight. * To evaluate the safety of once weekly injection of efpeglenatide.
Purpose
Efficacy and Safety of Efpeglenatide Versus Placebo in Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Basal Insulin Alone or in Combination With Oral Antidiabetic Drug(s)
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-11-09
Anticipated Date of Last Follow-up
2021-11-04
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-11-20
Actual Completion Date
2021-01-04
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion criteria: * Participant must be greater than or equal to (\>=)18 years of age at the time of signing the informed consent. * Participants with T2DM. * Diabetes diagnosed at least 1 year before screening. * Participants on basal insulin regimen alone or in combination with OADs for at least 6 months prior to screening. * HbA1c between 7.0 percent (%) and 10.0% (inclusive) measured by the central laboratory at screening. Exclusion criteria: * History of severe hypoglycemia requiring emergency room admission or hospitalization within 3 months prior to screening. * Retinopathy or maculopathy with one of the following treatments, either recent (within 3 months prior to screening) or planned: intravitreal injections or laser or vitrectomy surgery. * Clinically relevant history of ga
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
370
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-LIL2-101
Identifier
NCT06724016
Link
https://clinicaltrials.gov/study/NCT06724016
Phase
Phase I
Status
Recruiting
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This is a First-in-Human, Phase 1, Dose-Escalation and Dose-Expansion study of HM16390, as a single agent to assess safety, tolerability, MTD, RP2D, PK, and efficacy in patients with advanced or metastatic solid tumors. Dose-Escalation Part is planned to establish the MTD or RDs for the randomized Dose-Ranging Part. Based on the results of the Dose-Escalation Part, additional eligible subjects will be randomized 1:1 into at each dose level. After a comprehensive review of available data from both Dose-Escalation Part and Dose-Ranging Part, the potential RP2D to be tested in the Dose-Expansion Part is determined. Dose-Expansion Part is designed to assess the potential efficacy of HM16390 as a single agent when administered at the potential RP2D to subjects in indication-specific expansion
Purpose
Dose Escalation and Expansion Study of HM16390 in Advanced or Metastatic Solid Tumors
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
2024-12-11
Actual Start Date
Not provided
Anticipated Date of Last Follow-up
2024-12-11
Estimated Primary Completion Date
2030-03-01
Estimated Completion Date
2030-03-01
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Key Inclusion Criteria: * Have a histologically and/or cytologically confirmed advanced or metastatic solid tumor and have failed or are intolerant to standard therapy with clinical benefit. * Patients in the Dose-Escalation Part must have evaluable or measurable disease at baseline and the patients for Dose-Ranging and Dose-Expansion Part must have at least one measurable lesion at baseline by computed tomography (CT) or magnetic resonance imaging (MRI) per Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. * Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1. * Age of 18 years or older (or country's legal age of majority if the legal age was \>18 years) * Adequate renal function. * Adequate hematologic function. * Adequate liver function. Key Exclusion Crit
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
215
Allocation
Not provided
Intervention model
Single group assignment
Intervention model description
Not provided
Masking
Open label
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-101
Identifier
NCT03374241
Link
https://clinicaltrials.gov/study/NCT03374241
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
Single ascending dose of HM15211 in healthy obese subjects.
Purpose
A First-in-human Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of HM15211(Efocipegtrutide)
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-04-04
Anticipated Date of Last Follow-up
2025-01-23
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2018-09-14
Actual Completion Date
2018-09-14
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Female subjects must be non-pregnant and non-lactating Exclusion Criteria: * Participation in an investigational study within 30 days prior to dosing
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
41
Allocation
Randomized
Intervention model
Sequential assignment
Intervention model description
Not provided
Masking
Triple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-102
Identifier
NCT03744182
Link
https://clinicaltrials.gov/study/NCT03744182
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This study is a phase 1 study to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of multiple doses of HM15211 in obese subjects with NAFLD
Purpose
A Study of Multiple Doses of HM15211(Efocipegtrutide) in Obese Subjects With NAFLD
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-11-01
Anticipated Date of Last Follow-up
2025-01-23
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-03-18
Actual Completion Date
2020-03-18
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Body mass index ≥ 30 kg/m2 * Waist circumference ≤ 57 inches * Fasting Plasma Glucose \< 7 mmol/L (126 mg/dL) * HbA1c \< 6.5% * Controlled Attenuation Parameter ≥ 300 dB/m by FibroScan * Liver fat by MRI-PDFF ≥ 10%. Exclusion Criteria: * A history of or active chronic liver disease due to alcohol, auto-immune, HIV, HBV or active HCV-infection or NASH disease * Any history of clinically significant chronic liver disease including esophageal varices, ascites, encephalopathy or any hospitalization for treatment of chronic liver disease; or Model for End Stage Liver Disease (MELD) ≥ 10 * Previous surgical treatment for obesity * Uncontrolled hypertension * Any weight control treatment * History or current diagnosis of acute or chronic pancreatitis or factors for pancre
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
66
Allocation
Randomized
Intervention model
Sequential assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-TRIA-201
Identifier
NCT04505436
Link
https://clinicaltrials.gov/study/NCT04505436
Phase
Phase II
Status
Recruiting
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This study is a phase 2 study to Evaluate Efficacy, Safety and Tolerability of HM15211 Treatment for 12 Months in Subjects with Biopsy Confirmed NASH
Purpose
Study to Evaluate Efficacy, Safety and Tolerability of HM15211(efocipegtrutide) in Subjects
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2020-07-31
Anticipated Date of Last Follow-up
2025-01-20
Estimated Primary Completion Date
2025-05-11
Estimated Completion Date
2025-11-10
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: * United States Sites: Adults ≥ 18 to ≤ 70 years. * Korean Sites: Adults ≥ 19 to ≤ 70 years. * BMI ≥ 18 kg/m2, with stable body weight (defined as change \< 5%) by history for 3 months prior to screening or since baseline liver biopsy, whichever is earlier. * Subjects have a diagnosis of noncirrhotic NASH with liver fibrosis (Fibrosis stage F1-F3) confirmed by liver biopsy within 6 months of Day -7. * MRI-PDFF performed at screening with ≥ 8% steatosis. Exclusion Criteria: * Subjects with a history of active or chronic liver disease, including alcoholic liver disease, viral hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, autoimmune hepatitis, Wilson's disease, hemochromatosis, alpha-1 antitrypsin deficiency, human immunodeficiency virus (HIV). *
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
240
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-GHA-101
Identifier
NCT01093742
Link
https://clinicaltrials.gov/study/NCT01093742
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
* Study Design * Randomized, Double-blind, Placebo-controlled, escalating single-dose design. * Four ascending dose cohorts. * In each cohort, subjects will be randomized to receive a single dose of HM10560A or placebo (negative control). * Objectives * The primary objective of the study is to assess the safety and tolerability of single escalating subcutaneous doses of HM10560A in healthy male subjects.
Purpose
A Study of HM10560A in Healthy Male Subject
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2010-03-01
Anticipated Date of Last Follow-up
2014-02-06
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2010-08-01
Actual Completion Date
2010-08-01
Studied populations
Age Cohort
- Adults
Genders
- Male
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: 1. Healthy male volunteers, age range 20 to 54 years are informed of the investigational nature of this study and voluntarily agrees to participate in this study 2. Body mass index of ≥19 and ≤26 Subject 3. Medically healthy with no clinically significant screening results through Physical examination, 12 lead ECG, Laboratory test 4. Able to participate in all procedure 5. SBP 90-140 mmHg, DBP 60-90 mmHg, Pulse rate 50-90 times/min 6. AST, ALT \<1.5 X UNL, CPK \< 2 X UNL 7. Able to abstain from alcohol and smoke during study period 8. Consented to contraception until 2 month after end of the study Exclusion Criteria: 1. Acute infection history within 14 days 2. Prior exposure or hypersensitivity to recombinant human growth hormone 3. Positive findings on HBsAg, Anti-
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
32
Allocation
Randomized
Intervention model
Single group assignment
Intervention model description
Not provided
Masking
Triple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-GLP2-101
Identifier
NCT04076293
Link
https://clinicaltrials.gov/study/NCT04076293
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
A First-in-Human, Double-blind, Randomized, Placebo-controlled, Single Ascending Dose Study to Assess Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of HM15912 in Healthy Korean Subjects
Purpose
Single Ascending Dose Study to Assess Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of HM15912(Sonefpeglutide) in Healthy Korean Subjects
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2019-10-08
Anticipated Date of Last Follow-up
2025-01-23
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2020-11-10
Actual Completion Date
2021-05-03
Studied populations
Age Cohort
- Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Subject voluntarily agrees to participate in this study and signs an IRB-approved informed consent prior to performing any of the Screening visit procedures. * Korean males and females ≥ 19 and ≤ 60 years of age at the Screening visit Exclusion Criteria: * Subject with a history or presence of clinically significant active diseases. * Subject who has participated in other clinical studies (including bioequivalence tests) within 6 months before the Screening visit and has received IPs
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
40
Allocation
Randomized
Intervention model
Sequential assignment
Intervention model description
Not provided
Masking
Quadruple-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-GLP2-201
Identifier
NCT04775706
Link
https://clinicaltrials.gov/study/NCT04775706
Phase
Phase II
Status
Recruiting
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This is a randomized, double-blind, placebo-controlled, proof-of-concept (PoC), Phase 2 study to assess the safety, PK, and PD of SC administration of HM15912(sonefpeglutide) in adult subjects with SBS-associated intestinal failure (SBS-IF).
Purpose
Phase 2 Study to Assess the Safety, PK, and PD in SBS-IF Subjects
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2022-03-03
Anticipated Date of Last Follow-up
2025-02-06
Estimated Primary Completion Date
2027-12-01
Estimated Completion Date
2028-05-01
Actual Primary Completion Date
Not provided
Actual Completion Date
Not provided
Studied populations
Age Cohort
- Adults
- Older Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
No
Comments about the studied populations
Inclusion Criteria: 1. Men or women, aged 18 years of age or older with SBS resulting in intestinal failure at the time of signing the informed consent form (ICF) (or country's legal age of majority if the legal age is \<18 years) 2. Capable of giving signed informed consent which includes compliance with the requirements and restrictions listed in the ICF and in this protocol 3. Diagnosis of SBS with the latest intestinal resection being at least 6 months prior to Screening and considered stable regarding the PN/IV need. No restorative surgery planned in the study period. Exclusion Criteria: 1. Any history of colon cancer. 2. History of any other cancers (except margin-free resected cutaneous basal or squamous cell carcinoma or adequately treated in situ cervical cancer) unless disease
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
18
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
HM-GCG-101
Identifier
NCT04032782
Link
https://clinicaltrials.gov/study/NCT04032782
Phase
Phase I
Status
Completed
Sponsor
Hanmi Pharmaceutical Company Limited
More details
This is a double-blind, randomized, placebo controlled, single ascending dose (SAD) study to investigate the safety, tolerability, PK and PD of the SC administration of HM15136 in healthy subjects. The study will be conducted in approximately 5 sequential dosing cohorts, enrolling 8 subjects per cohort. Subjects will be randomized to HM15136 or placebo in a ratio of 6:2 (6 active, 2 placebo).
Purpose
A First-in-Human, Study to Assess Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of HM15136(Efpegerglucagon) in Healthy Subjects
Interventions
Not provided
Countries
Not provided
Sites / Institutions
Not provided
Trials dates
Anticipated Start Date
Not provided
Actual Start Date
2018-10-02
Anticipated Date of Last Follow-up
2025-02-03
Estimated Primary Completion Date
Not provided
Estimated Completion Date
Not provided
Actual Primary Completion Date
2019-08-29
Actual Completion Date
2019-08-29
Studied populations
Age Cohort
- Adults
Genders
- All
Accepts pregnant individuals
Unspecified
Accepts lactating individuals
Unspecified
Accepts healthy individuals
Yes
Comments about the studied populations
Inclusion Criteria: * Body mass index ≥ 18.5 and ≤ 27 kg/m2 and with a weight ≥ 50 kg Exclusion Criteria: * with personal or family history of hypercoagulability or thromboembolic disease * has had treatment with any incretin therapy * has FPG \< 70 or \> 110 mg/dL
Health status
Not provided
Study type
Interventional (clinical trial)
Enrollment
40
Allocation
Randomized
Intervention model
Parallel Assignment
Intervention model description
Not provided
Masking
Double-blind masking
Masking description
Not provided
Frequency of administration
Not provided
Studied LA-formulation(s)
Not provided
Studied route(s) of administration
Not provided
Use case
Not provided
Key resources
Excipients
Proprietary excipients used
No proprietary excipient used
Novel excipients or existing excipients at a concentration above Inactive Ingredients Database (IID) for the specified route of administration
No novel excipient or existing excipient used
Residual solvents used
No residual solvent used
Additional features
Other features of the technology
- Drug-eluting
- At least 1 year shelf life
Release properties
Upon reaching the target site, the aglycosylated Fc-bound API is released via the detachment of the flexible PEG linker. Once freed, the API binds to its target receptor, eliciting its therapeutic effect through agonistic or antagonistic activity. This monomeric complex partially reduces receptor-mediated clearance and renal filtration. Due to this mechanism, the activity of the API is prolonged.
Injectability
The prefilled injection of efpeglenatide (LAPScovery formulation) typically uses a 29-gauge needle for subcutaneous administration.
Safety
In the phase 3 clinical trial evaluating once-weekly efpeglenatide, the most frequently reported adverse events were mild to moderate gastrointestinal symptoms, including diarrhea, vomiting, and constipation. Treatment-emergent adverse events (TEAEs) occurred in 78.4–83.8% of patients in the treatment group, with two cases of diabetic retinopathy being reported. Serious TEAEs were observed in 9–11% of the patient population.
Stability
LASPSCOVERY formulation in its prefilled pen or vial is stable for up to 24 months when stored under the recommended refrigeration condition
Storage conditions and cold-chain related features
Store in a refrigerator at 2°C to 8°C (36°F to 46°F). Do not freeze. It can be stored at room temperature (up to 30°C/86°F) for a maximum of 14 days.
Potential application(s)
Therapeutic area(s)
Use case(s)
Not provided
Use of technology
Ease of administration
- Administered by a community health worker
- Administered by a nurse
- Administered by a specialty health worker
Frequency of administration
Weekly, Monthly
User acceptance
Not provided
Targeted user groups
Age Cohort- Adults
- Older Adults
- All
Pregnant individuals
Unspecified
Lactating individuals
Unspecified
Healthy individuals
Unspecified
Comment
Not provided
Potential associated API(s)
efpegerglucagon (glucagon analog)
Class(es)
Glucagon Analog
Development stage
Phase II
Clinical trial number(s)
NCT04732416
Foreseen/approved indication(s)
Congenital Hyperinsulinism (CHI)
Foreseen user group
2 years and older
Foreseen duration between application(s)
Once weekly
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Efpegerglucagon received orphan drug designation from the FDA, EMA, and MFDS for the treatment of CHI
efpeglenatide (exendin-4 analog)
Class(es)
Exendin-4 Analog
Development stage
Phase III
Clinical trial number(s)
NCT03353350
Foreseen/approved indication(s)
Type 2 Diabetes
Foreseen user group
< 18 years and older
Foreseen duration between application(s)
Once weekly
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Not provided
efocipegtrutide (GLP-1 triple agonist )
Class(es)
GLP-1 triple agonist
Development stage
Phase II
Clinical trial number(s)
NCT04505436
Foreseen/approved indication(s)
nonalcoholic steatohepatitis (MASH)
Foreseen user group
Adults ≥ 18 to ≤ 70 years
Foreseen duration between application(s)
Once weekly
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Efocipegtrutide Fast Track designation and orphan drug status for the treatment of nonalcoholic steatohepatitis (MASH).
HM16390 (IL-2 analog)
Class(es)
Interleukin Analogues
Development stage
Phase I
Clinical trial number(s)
NCT06724016
Foreseen/approved indication(s)
Advanced or Metastatic Solid Tumors
Foreseen user group
Not provided
Foreseen duration between application(s)
Not provided
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Not provided
efpegsomatropin (human growth hormone (hGh) analog)
Class(es)
Somatotropin receptor agonists
Development stage
Phase II
Clinical trial number(s)
Not provided
Foreseen/approved indication(s)
Somatotropin deficiency
Foreseen user group
Not provided
Foreseen duration between application(s)
Not provided
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Not provided
sonefpeglutide (Glucagon-like peptide-2 (GLP-2) analogue)
Class(es)
GLP-2 analog
Development stage
Phase II
Clinical trial number(s)
NCT04775706
Foreseen/approved indication(s)
Short Bowel Syndrome-associated Intestinal Failure
Foreseen user group
Not provided
Foreseen duration between application(s)
Once monthly
Applications to Stringent Regulatory Authorities (SRA) / regulatory approvals
Not provided
Patent info
Description
Composition for treating diabetes comprising long-acting insulin conjugate and long-acting insulinotropic peptide conjugate
Brief description
The present invention relates to an anti-cancer pharmaceutical composition comprising an interferon alpha or a polymer congugate thereof and use thereof in the treatment of cancer by co-administration with anti-cancer agents. The interferon alpha conjugate of the present invention shows a longer in vivo half-life and a more excellent anti-cancer activity than the conventional interferon alpha, and in particular, its co-administration with an anti-cancer agent such as gemcitabine has synergistic inhibitory effects on cancer cell growth and proliferation so as to exhibit a remarkably excellent anti-cancer activity. Further, the anti-cancer pharmaceutical composition of the present invention has excellent in vivo half-life and anti-cancer activity to greatly reduce administration frequency. C
Representative patent
WO2013036032
Category
Formulation
Patent holder
Hanmi Science Co Ltd
Exclusivity
Not provided
Expiration date
June 1, 2032
Status
Granted: AU, EP (BE, CH, FR, IT, LI), JP, MX, RU, US Pending: CA, CN Not in force: BR
Description
Long-acting glucagon conjugate and pharmaceutical composition comprising the same for the prevention and treatment of obesity
Brief description
Disclosed is a novel long-acting glucagon conjugate in which glucagon or its derivative is covalently linked to a polymer carrier via a non-peptide linker, and a pharmaceutical composition comprising the same as an effective ingredient useful for the prevention and treatment of obesity. Since the long-acting glucagon conjugate of the present invention shows improved in vivo durability and stability, when used in combination with an anti-obesity drug, it is possible to induce synergistic effects on the loss of body weight and decrease in food intake without causing any side-effects such as fluctuation in blood glucose level. Accordingly, the long-acting peptide conjugate of the present invention is very effective for the prevention and treatment of obesity.
Representative patent
WO2012011752
Category
Compound
Patent holder
Hanmi Science Co Ltd
Exclusivity
Not provided
Expiration date
September 21, 2031
Status
Granted: EP (DE, FR, GB), JP, KR, US Pending: AR
Description
Protein conjugate comprising polypetide drug, aglycosylated IgG4 Fc fragment, and PEG linker
Brief description
Disclosed are a protein conjugate with improved in vivo duration and stability and the use thereof. The protein conjugate includes a physiologically active polypeptide, a non-peptide polymer and an immunoglobulin Fc fragment. Since the three components are covalently linked, the protein conjugate has extended in vivo duration and enhanced stability for the physiologically active polypeptide. The protein conjugate maintains the in vivo activity at relatively high levels and remarkably increases the serum half-life for the physiologically active polypeptide, with less risk of inducing undesirable immune responses. Thus, the protein conjugate is useful for developing long-acting formulations of various polypeptide drugs.
Representative patent
WO2005047336
Category
Compound
Patent holder
HANMI PHARMACEUTICAL. CO., LTD.
Exclusivity
Not provided
Expiration date
November 13, 2024
Status
Expired
Supporting material
Publications
<p><span style="color: rgb(33, 33, 33);">Choi, J., Lee, J., Park, E., Kwon, H., Kim, D., Bae, S., Choi, I. Y., & Kim, H. H. (2023). HM15912, a Novel Long-Acting Glucagon-Like Peptide-2 Analog, Improves Intestinal Growth and Absorption Capacity in a Male Rat Model of Short Bowel Syndrome. </span><em style="color: rgb(33, 33, 33);">The Journal of pharmacology and experimental therapeutics</em><span style="color: rgb(33, 33, 33);">, </span><em style="color: rgb(33, 33, 33);">384</em><span style="color: rgb(33, 33, 33);">(2), 277–286. </span><a href="https://doi.org/10.1124/jpet.122.001381" rel="noopener noreferrer" target="_blank" style="color: rgb(33, 33, 33);">https://doi.org/10.1124/jpet.122.001381</a></p>
Extensive bowel resection caused by various diseases that affect the intestines, such as Crohn's disease, volvulus, and cancer, leads to short bowel syndrome (SBS). Teduglutide is the only approved glucagon-like peptide-2 (GLP-2) drug for SBS; however, it requires daily administration. A novel GLP-2 analog with a prolonged duration of action to reduce dosing frequency and promote a greater efficacy may provide patients with a better quality of life. In the present study, the sustained exposure of HM15912 was characterized in normal male rats. The efficacy of HM15912 on intestinal growth and absorption capacity was also evaluated in normal male mice, rats, and SBS rats. HM15912 exhibited a remarkably extended half-life (42.3 hours) compared with teduglutide (0.6 hours) in rats. Despite somewhat lower in vitro potency on GLP-2 receptor than human GLP-2 or teduglutide, this longer-lasting mode of action promotes HM15912 to be more effective in terms of small intestinal growth than existing GLP-2 analogs even with a less frequent dosing interval of as little as once a week in rodents, including SBS rats. Furthermore, the small intestinal weight was approximately doubled, and the D-xylose absorption was significantly increased after pre-treatment of existing GLP-2 analogs on the market or under clinical development followed by HM15912 in rodents. These results indicate that HM15912 possesses a significant small bowel trophic effect driven by continuously increased exposure, supporting that HM15912 may be a novel treatment option with greater efficacy and the longest dosing interval among existing GLP-2 analogs for SBS with intestinal failure. SIGNIFICANCE STATEMENT: HM15912, a novel long-acting glucagon-like peptide-2 (GLP-2) analog, has a significant small bowel hypertrophic effect in rodents with a reduced frequency of administration compared to the existing GLP-2 analogs on the market or currently under clinical development. This study supports the possibility that HM15912 could be administered much less frequently than other long-acting GLP-2 analogs for patients with short bowel syndrome.
Additional documents
Useful links
There are no additional links
Access principles
|
|
Collaborate for developmentConsider on a case by case basis, collaborating on developing long acting products with potential significant public health impact, especially for low- and middle-income countries (LMICs), utilising the referred to long-acting technology Not provided |
|
|
Share technical information for match-making assessmentProvide necessary technical information to a potential partner, under confidentiality agreement, to enable preliminary assessment of whether specific medicines of public health importance in LMICs might be compatible with the referred to long-acting technology to achieve a public health benefit Not provided |
|
|
Work with MPP to expand access in LMICsIn the event that a product using the referred to long-acting technology is successfully developed, the technology IP holder(s) will work with the Medicines Patent Pool towards putting in place the most appropriate strategy for timely and affordable access in low and middle-income countries, including through licensing Not provided |
Comment & Information
Illustrations

API binded with the Aglycosylated Fc fragment via a Flexible PEG linker
Hanmi Pharm. (n.d.). LAPScovery™: Long-Acting Pharmaceutical Solutions. Retrieved February 18, 2025, from https://hanmipharm.com
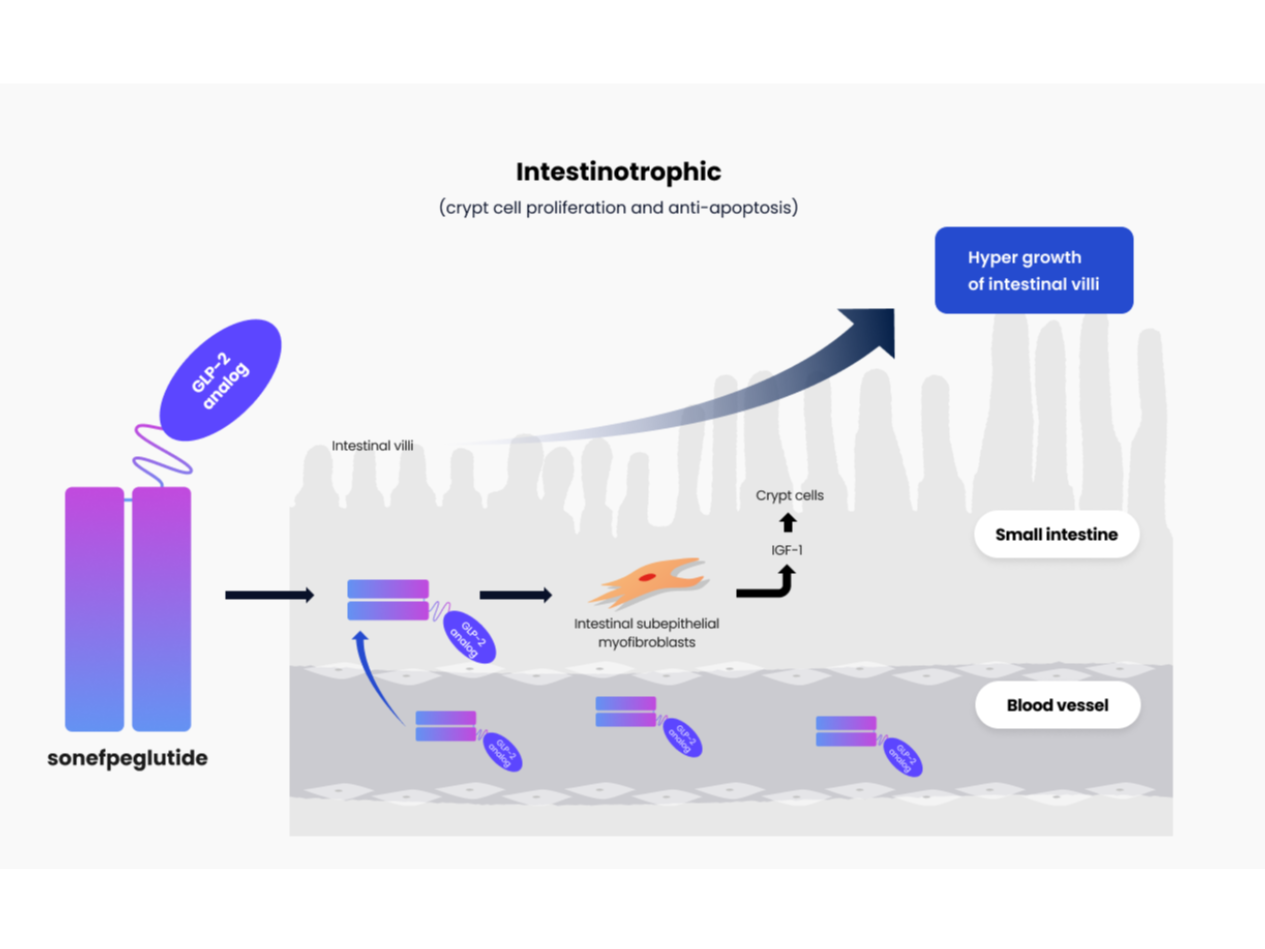
Mechanism of Action of Sonefpeglutide in the intestinal villi
Hanmi Pharmaceutical. (n.d.). HM15912: Weekly GLP-1/glucagon dual agonist. Retrieved [Date], from https://hanmipharm.com/science/pipeline/focused/hm15912.hm